SLIDE 1
Synthetic Polymers
The initiator can be a radical, a cation, or an anion. Mechanism for Radical Polymerization:
Ph H2O2 Ph
n
polystyrene (styrofoam)
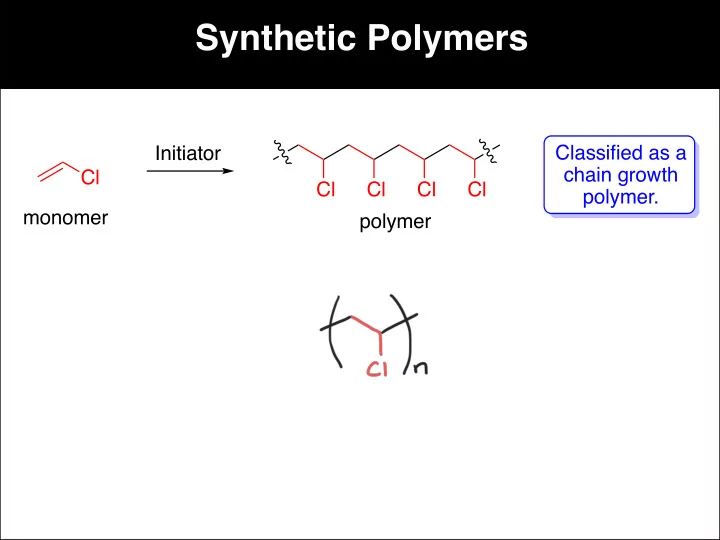
Synthetic Polymers Classified as a Initiator chain growth Cl Cl - - PowerPoint PPT Presentation
Synthetic Polymers Classified as a Initiator chain growth Cl Cl Cl Cl Cl polymer. monomer polymer Synthetic Polymers The initiator can be a radical, a cation, or an anion. Mechanism for Radical Polymerization: Ph H 2 O 2 polystyrene
Ph H2O2 Ph
n
polystyrene (styrofoam)