Rank the following solutions in order of increasing pH: 0.1 M HNO 2 - PDF document
Rank the following solutions in order of increasing pH: 0.1 M HNO 2 , 0.1 M HNO 3 , 0.1 M NaNO 2 , 0.1 M NaNO 3 , 0.1 M NaOH A. HNO 2 < HNO 3 < NaNO 2 < NaNO 3 < NaOH B. HNO 3 < HNO 2 < NaNO 2 < NaNO 3 < NaOH C. HNO 2 <
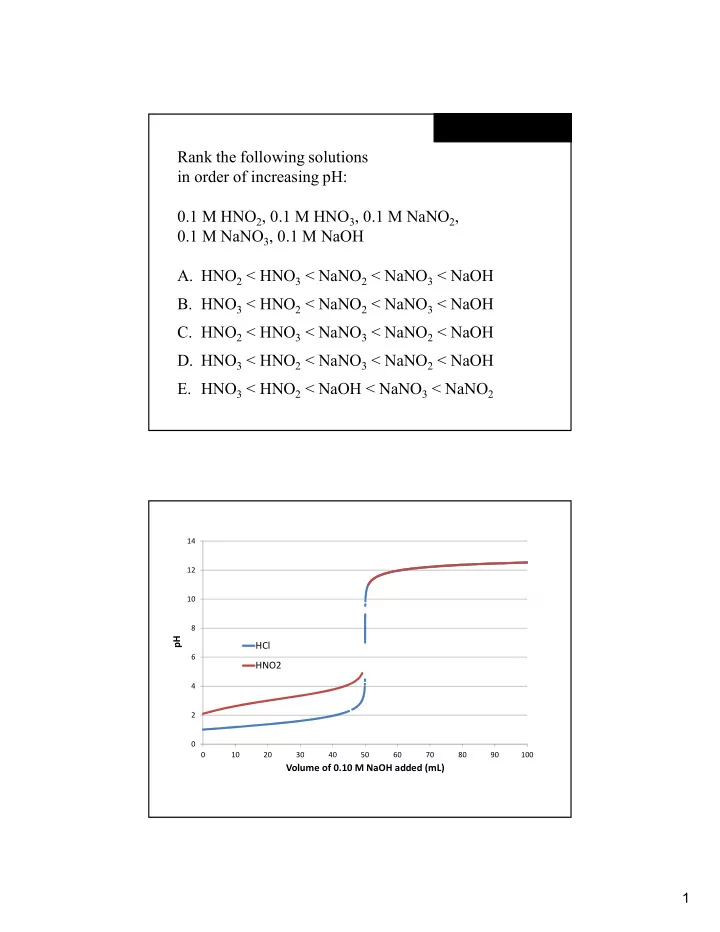
Rank the following solutions in order of increasing pH: 0.1 M HNO 2 , 0.1 M HNO 3 , 0.1 M NaNO 2 , 0.1 M NaNO 3 , 0.1 M NaOH A. HNO 2 < HNO 3 < NaNO 2 < NaNO 3 < NaOH B. HNO 3 < HNO 2 < NaNO 2 < NaNO 3 < NaOH C. HNO 2 < HNO 3 < NaNO 3 < NaNO 2 < NaOH D. HNO 3 < HNO 2 < NaNO 3 < NaNO 2 < NaOH E. HNO 3 < HNO 2 < NaOH < NaNO 3 < NaNO 2 14 12 10 8 pH HCl 6 HNO2 4 2 0 0 10 20 30 40 50 60 70 80 90 100 Volume of 0.10 M NaOH added (mL) 1
14 48 4.73 13 49 5.04 12 49.9 6.05 65 12.08 11 49.99 7.04 70 12.18 10 49.999 7.82 75 12.26 9 50 8.01 100 12.48 8 pH 7 50.001 8.20 1000 12.90 6 50.01 8.97 10000 12.94 5 50.1 9.97 4 0 2.19 51 10.96 3 52 11.26 2 1 0 0 10 20 30 40 50 60 70 80 90 100 110 volume of 0.1 M NaOH(aq) added 2
You can calculate the pH of 0.10 M NH 4 Cl( aq ) using 2 x the equation given on the K right where K and x are 0.10 x A. K a [H + ] [OH ] B. K a C. K b [H + ] [OH ] D. K b 3
4
m = 1 2 + c 2 z 2 2 +××× ) 1 2 å 2 2 ( c 1 z 1 i c i z i Ionic strength ( m ) is given by the equation where c i is the concentration of ion i, and z i is the charge of ion i . 5
6
is acid or basic, To determine if 0.10 M HCO 3 against the K b you must compare K a2 for HCO 3 which is given by for HCO 3 A. K b = K w /K a1 B. K b = K w /K a2 C. K b = K a1 /K w D. K b = K a2 /K w 7
H 2 CO 3 (aq) + OH − (aq) −−> HCO 3 − (aq) + OH − (aq) −−> CO 3 2− (aq) + H 2 O( l ) + H 2 O( l ) H 2 SO 3 (aq) + OH − (aq) −−> HSO 3 − (aq) + OH − (aq) −−> SO 3 2− (aq) + H 2 O( l ) + H 2 O( l ) 8
Recall… H 2 SO 3 (aq) + OH − (aq) −−> HSO 3 − (aq) + OH − (aq) −−> SO 3 2− (aq) + H 2 O( l ) + H 2 O( l ) 9
K a1 = 1.4 10 2 K a2 = 6.5 10 8 H 2 SO 3 HSO 3 SO 3 2 K a1 = 1.0 10 4 K a2 = 1.0 10 8 H 2 A HA A 2 10
K a1 = 1.0 10 5 K a2 = 1.0 10 7 H 2 A HA A 2 K a1 = 1.0 10 6 K a2 = 1.0 10 6 11
12
m = 1 2 + c 2 z 2 2 +××× ) 1 2 å 2 2 ( c 1 z 1 i c i z i Ionic strength ( m ) is given by the equation where c i is the concentration of ion i, and z i is the charge of ion i . Recall… H 2 SO 3 (aq) + OH − (aq) −−> HSO 3 − (aq) + OH − (aq) −−> SO 3 2− (aq) + H 2 O( l ) + H 2 O( l ) 13
K a1 = 1.4 10 2 K a2 = 6.5 10 8 H 2 SO 3 HSO 3 SO 3 2 K a1 = 1.0 10 4 K a2 = 1.0 10 8 H 2 A HA A 2 14
K a1 = 1.0 10 5 K a2 = 1.0 10 7 H 2 A HA A 2 K a1 = 1.0 10 6 K a2 = 1.0 10 6 15
0.46 V Cu 2+ ions → - ions ← NO 3 copper wire silver wire 1 M AgNO 3 1 M Cu(NO 3 ) 2 copper metal is oxidized at the anode: silver ions are reduced at the cathode: Cu(s) → Cu 2+ (aq) + 2e ‒ Ag + (aq) + e ‒ → Ag(s) 16
Cu(s) Ag(s) 0 4 6 1 M Cu 2+ 1 M Ag + Zn(s) Ag(s) 1 5 6 1 M Zn 2+ 1 M Ag + 17
Ag + ( aq ) + 1 e Ag( s ) 0.46 V Cu 2+ ( aq ) + 2 e Cu( s ) 1.56 V Zn 2+ ( aq ) + 2 e Zn( s ) Ag + ( aq ) + 1 e Ag( s ) 0.46 V Cu 2+ ( aq ) + 2 e Cu( s ) 1.56 V 1.10 V Zn 2+ ( aq ) + 2 e Zn( s ) 18
Zn(s) Cu(s) 1 1 0 1 M Zn 2+ 1 M Cu 2+ Ag + ( aq ) + 1 e Ag( s ) 0.36 V Cu 2+ ( aq ) + 2 e Cu( s ) 1.56 V 2H + ( aq ) + 2 e H 2 ( g ) 1.10 V Zn 2+ ( aq ) + 2 e Zn( s ) 19
Ag + ( aq ) + 1 e Ag( s ) 0.36 V Cu 2+ ( aq ) + 2 e Cu( s ) 1.56 V 2H + ( aq ) + 2 e H 2 ( g ) 0.000 V 1.10 V Zn 2+ ( aq ) + 2 e Zn( s ) Ag + ( aq ) + 1 e Ag( s ) 0.80 V 0.46 V Cu 2+ ( aq ) + 2 e Cu( s ) 0.34 V 1.56 V 2H + ( aq ) + 2 e H 2 ( g ) 0.000 V 1.10 V Zn 2+ ( aq ) + 2 e Zn( s ) 0.76 V 20
E°(V) Ag + ( aq ) + 1 e Ag( s ) 0.80 Cu 2+ ( aq ) + 2 e Cu( s ) 0.34 2H + ( aq ) + 2 e H 2 ( g ) 0.000 Zn 2+ ( aq ) + 2 e Zn( s ) 0.76 21
22 Decreasing potential wants electrons less Decreasing potential wants electrons less weaker oxidizing agent product is a weaker oxidizing agent product is a stronger reducing agent stronger reducing agent stronger oxidizing agent stronger oxidizing agent Increasing potential wants electrons more Increasing potential wants electrons more
23
Recommend
More recommend
Explore More Topics
Stay informed with curated content and fresh updates.























