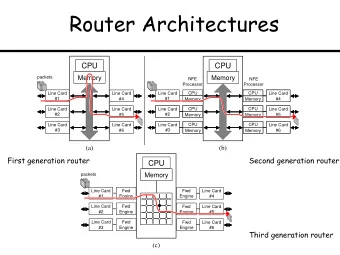
Top-Line Data Results Omadacycline in Acute Skin and Skin Structure - PowerPoint PPT Presentation
ABSSSI-16301 Study Design: Top-Line Data Results Omadacycline in Acute Skin and Skin Structure Infections Study (OASIS-2) A Phase 3 Randomized, Double-Blind, Multi-Center Study to Compare the Safety and Efficacy of Oral Omadacycline to
ABSSSI-16301 Study Design: Top-Line Data Results Omadacycline in Acute Skin and Skin Structure Infections Study (OASIS-2) A Phase 3 Randomized, Double-Blind, Multi-Center Study to Compare the Safety and Efficacy of Oral Omadacycline to Linezolid for Treating Adult Subjects with ABSSSI 17 July 2017 1
Safe Harbor Statement Certain statements in this presentation, including responses to questions, contain or may contain “forward - looking statements” w ithin the meaning of the Private Securities Litigation Reform Act of 1995. Examples of such statements include, but are not limited to, statements about our strategy, future operations, prospects, plans, objectives of management, availability of data from our clinical studies, potential use of our product candidates, including Omadacycline and Sarecycline, the market acceptance of our product candidates, the strength of, and protection offered by, our intellectual property position, the potential clinical risks and efficacy of, and market opportunities for, our product candidates, the timing and stability of our supply chain, the timing of clinical development of, and regulatory approval for, our product candidates, and the nature and timing of our collaboration agreements with respect to our product candidates. The words “anticipate,” “estimate,” “expect,” “potential,” “ wil l,” “project” and similar terms and phrases are used to identify forward-looking statements. These statements are based on current information and belief and are not guarantees of future performance. Our ability to predict results, financial or otherwise, or the actual effect of future plans or strategies, is inherently uncertain and actual results may differ from those predicted depending on a variety of factors. Our operations involve risks and uncertainties, many of which are outside our control, and any one of which, or a combination of which, could materially affect our results of operations or whether the forward-looking statements ultimately prove to be correct. Except as required by law, we undertake no obligation to publicly update any forward- looking statement, whether as a result of new information, future events or otherwise. Among the risks and uncertainties that could cause actual results to differ materially from those indicated by such forward-looking statements include: delays in clinical trials or unexpected results; the risk that data to date and trends may not be predictive of future results; the failure of collaborators to perform obligations under our collaboration agreements; our failure to obtain regulatory approval for our product candidates; if we obtain regulatory approval for our product candidates, the risk that the terms of such approval may limit how we manufacture and market our product candidates; delays in our supply chain, delays in undertaking or completing clinical trials; our products not gaining the anticipated acceptance in the marketplace or acceptance being delayed; our products not receiving reimbursement from healthcare payors; the effects of competition; our inability to protect our intellectual property and proprietary technology through patents and other means; the need for substantial additional funding to complete the development and commercialization of our product candidates; and the other risks described in the “Risk Factors” section and elsewhere in our Annual Report on Form 10-K for the year ended December 31, 2016, and our other filings with the SEC. 2
ABSSSI-16301 Study Design: Top-Line Data Results Omadacycline in Acute Skin and Skin Structure Infections Study (OASIS-2) Michael Bigham CEO and Chairman of the Board Evan Loh, MD President, COO, and CMO 3
Acute Bacterial Skin and Skin Structure Infections Unmet need for antibiotics (especially oral options) to treat serious ABSSSI infections – Hospital admissions up 73% between 1997-2011 – Average hospital stay 5.2 days at approx. $10,000 / stay – Tetracyclines (e.g., oral doxycycline) emerged as important options for ABSSSI caused by MRSA, however, use is limited by resistance Suture abscess and cellulitis Omadacycline has potential to meet this need – Potent activity against most common ABSSSI pathogens (including MSSA/MRSA, Group A Strep) – Oral and IV option – Circumvents common tetracycline resistance mechanisms – Important treatment option for patients who can’t receive other antimicrobial classes (e.g., intolerance or bacterial Abscess on the resistance leading to poor response) back Cellulitis 4
Phase 3 Trial Design for OASIS-2 (ABSI-16301) Designed to Satisfy FDA and EMA Requirements Oral Omadacycline ABSSSI 735 patients [1] Oral Linezolid d1 d2-3 d7 to d14 7-14d after last treatment day End of Treatment FDA - Early Clinical EMA - Post-Treatment Response [2] Evaluation [3] [1] 720 mITT Subjects with qualifying infections [2] Early Clinical Response = primary end point for FDA. [3] PTE end point = Co-primary end points confirmed through EMA scientific advice . Day 1 Day 2 Day 3 and beyond 0h 12h 24h 36h 450 mg 450 mg Omadacycline After Day 2: 300mg oral once daily Oral Oral 600mg 600mg 600mg 600mg Linezolid 600mg oral twice daily Oral Oral Oral Oral 5
Demographics, Baseline Characteristics, and Subject Disposition
Key Study Populations Balanced Between Treatment Arms Omadacycline Linezolid All subjects Population n (%) n (%) n (%) ITT 368 367 735 Safety 368 (100.0) 367 (100.0) 735 (100.0) mITT 360 (97.8) 360 (98.1) 720 (98.0) micro-mITT 276 (75.0) 287 (78.2) 563 (76.6) CE-PTE 284 (77.2) 292 (79.6) 576 (78.4) ITT: all subjects who were randomized Safety: subjects who received test article mITT: randomized subjects without a sole Gram-negative causative pathogen at screening micro-mITT: Consists of subjects in mITT with at least one Gram-positive causative bacterial pathogen CE-PTE: mITT subjects who received test article, had a qualifying ABSSSI, an assessment of outcome at PTE, and met other key evaluability criteria Source: 14.1.1.2 7
Safety Population Demographics Balanced Between Treatment Arms Omadacycline Linezolid All Subjects Characteristics (n=368) (n=367) (n=735) Gender n (%) Female 126 (34.2) 147 (40.1) 273 (37.1) Male 242 (65.8) 220 (59.9) 462 (62.9) Age (years) Mean (SD) 42.8 (12.72) 44.5 (13.11) 43.7 (12.94) Median 41.0 46.0 43.0 Min, Max 18, 86 20, 84 18, 86 Categorical Age (years) n (%) 18-45 213 (57.9) 183 (49.9) 396 (53.9) >45-65 141 (38.3) 164 (44.7) 305 (41.5) >65-75 11 (3.0) 12 (3.3) 23 (3.1) >75 3 (0.8) 8 (2.2) 11 (1.5) Weight (kg) n 368 367 735 Mean (SD) 81.62 (18.286) 80.15 (19.778) 80.89 (19.047) Median 79.40 76.20 77.70 Min, Max 41.7, 167.0 44.5, 156.3 41.7, 167.0 BMI (kg/m^2) n 368 367 735 Mean (SD) 27.91 (6.472) 27.93 (6.556) 27.92 (6.510) Source: 14.1.2.1 8
Type of Primary Infection Balanced Between Treatment Arms Omadacycline Linezolid (n=360) (n=360) Characteristics n (%) n (%) Type of Primary Infection 360 360 Wound Infection 210 (58.3) 214 (59.4) Cellulitis/erysipelas 86 (23.9) 84 (23.3) Major Abscess 64 (17.8) 62 (17.2) Source: 14.1.4.1.1 9
Subject Disposition – ITT Population (Completed Study Treatment) High Percentage of Patients Completing Treatment Omadacycline Linezolid All Subjects (n=368) (n=367) (n=735) Parameter/Category n (%) n (%) n (%) Randomized 368 (100.0) 367 (100.0) 735 (100.0) Completed Study Treatment 328 (89.1) 315 (85.8) 643 (87.5) Prematurely Discontinued from Study Treatment 40 (10.9) 52 (14.2) 92 (12.5) Reason For Premature Discontinuation from Study Treatment Adverse Event 6 (1.6) 4 (1.1) 10 (1.4) Lost to Follow-up 18 (4.9) 25 (6.8) 43 (5.9) Withdrawal by Subject 6 (1.6) 8 (2.2) 14 (1.9) Physician Decision 3 (0.8) 7 (1.9) 10 (1.4) Death 0 0 0 Other 7 (1.9) 8 (2.2) 15 (2.0) Source: 14.1.1.4.1 10
Efficacy Results
Primary Endpoints Achieved for Both FDA and EMA Omadacycline Linezolid 97.9 95.5 100 87.5 84.2 90 82.5 80.8 80 Clinical Success 70 60 (%) 50 40 30 20 10 0 mITT Early Clinical Response mITT PTE - Clinical Success CE-PTE - Clinical Success Delta (95% CI) Delta (95% CI) Delta (95% CI) +5.0 (-0.2, 10.3) +3.3 (-2.2, 9.0) +2.3 (-0.5, 5.8) FDA Primary Endpoint EMA Co-Primary Endpoints Source: 14.2.1.1.1 and 14.2.2.1.1 12
Clinical Success at PTE by Gram-Positive Baseline Pathogen Highly Effective Across Key Pathogens Omadacycline Linezolid (n=276) (n=287) Clinical Success Clinical Success Baseline Pathogen n1 n (%) n1 n (%) Staphylococcus aureus 220 182 (82.7) 233 186 (79.8) MRSA 104 89 (85.6) 107 85 (79.4) MSSA 120 97 (80.8) 130 103 (79.2) Staphylococcus lugdunensis 5 4 (80.0) 0 0 Streptococcus pyogenes 29 20 (69.0) 16 9 (56.3) Streptococcus anginosus group 57 49 (86.0) 45 33 (73.3) Streptococcus anginosus 27 24 (88.9) 20 16 (80.0) Streptococcus intermedius 23 18 (78.3) 24 16 (66.7) Streptococcus constellatus 9 8 (88.9) 7 5 (71.4) Enterococcus faecalis 8 8 (100.0) 12 9 (75.0) VRE 0 0 2 2 (100.0) VSE 7 7 (100.0) 10 7 (70.0) Source: 14.2.2.6.1 13
Safety Results
Recommend
More recommend
Explore More Topics
Stay informed with curated content and fresh updates.