Physics 115
General Physics II Session 13
Specific heats revisited Entropy
4/22/14 Physics 115 1
- R. J. Wilkes
- Email: phy115a@u.washington.edu
- Home page: http://courses.washington.edu/phy115a/
Physics 115 General Physics II Session 13 Specific heats revisited - - PowerPoint PPT Presentation
Physics 115 General Physics II Session 13 Specific heats revisited Entropy R. J. Wilkes Email: phy115a@u.washington.edu Home page: http://courses.washington.edu/phy115a/ 4/22/14 Physics 115 1 Lecture Schedule (up to exam 2)
4/22/14 Physics 115 1
4/22/14 Physics 115
2
4/22/14 Physics 115 3
4/22/14 Physics 115 4
4/22/14 Physics 115 5
Adiabatic compression Adiabatic expansion
Isotherm
4/22/14 Physics 115 6
4/22/14 Physics 115 7
4/22/14 Physics 115 8
4/22/14 Physics 115 9
4/22/14 Physics 115 10
4/22/14 Physics 115 11
U = 3
2 nRT
CV = ΔU mol• K = 3
2 R
CP = CV + R = 5
2 R
4/22/14 Physics 115 12
i = nRTi Vi = 2.5mol 8.31 J/mol K
iVi 5 3 = PfVf 5 3
i Vi Vf
5 3 = 104.7kPa
4/22/14 Physics 115 13
4/22/14 Physics 115 14
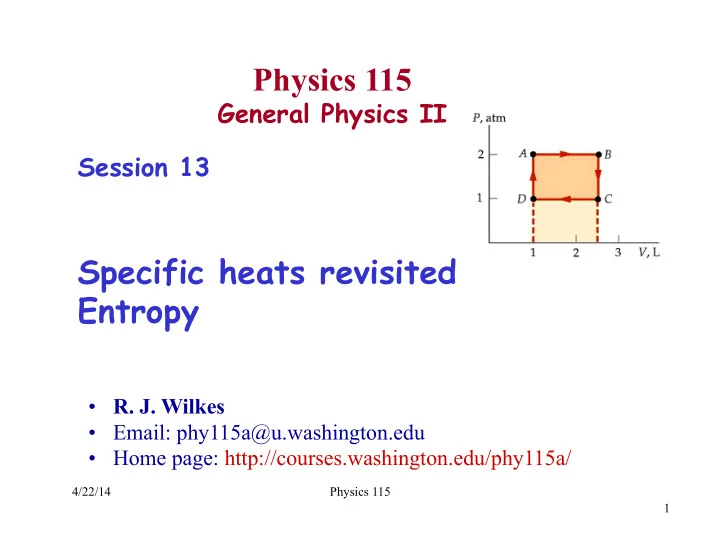
WAB = PΔV = (2.00 atm)(2.5 L−1.0 L) = 3.00 L⋅atm = 304 J WBC =WDA = 0 WCD = PΔV = (1.00 atm)(1.0 L− 2.5 L) = −1.50 L⋅atm = −152 J WNET, ON = −WCD −WAB = −152 J (more work done by gas than on gas)
4/22/14 Physics 115 15
4/22/14 Physics 115 16
4/22/14 Physics 115 17
4/22/14 Physics 115 18